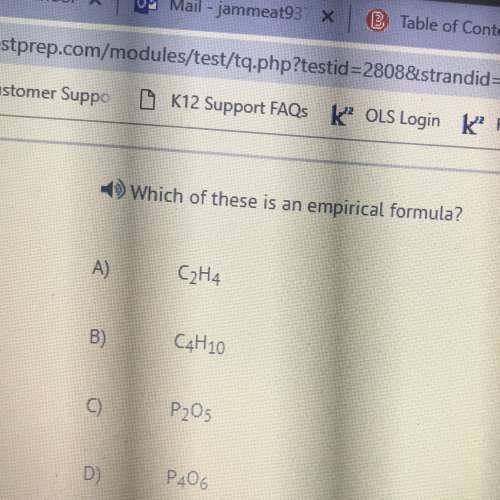
Chemistry, 05.06.2020 23:00 journeewilliams735
Can someone help me with this chemistry question? (This isn't the question but I need some help, I'll give 50 points to whoever can correctly answer my next question)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:30
Modern weaponry has increased the number of deaths in wars and violent conflicts.
Answers: 3

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 19:30
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2

Chemistry, 22.06.2019 21:00
Which property of water causes water drops to bead on a freshly waxed car?
Answers: 2
You know the right answer?
Can someone help me with this chemistry question?
(This isn't the question but I need some help, I'...
Questions


Mathematics, 22.11.2020 06:40

Mathematics, 22.11.2020 06:40

Physics, 22.11.2020 06:40


Social Studies, 22.11.2020 06:40

Social Studies, 22.11.2020 06:40

English, 22.11.2020 06:40

English, 22.11.2020 06:40

Advanced Placement (AP), 22.11.2020 06:40

History, 22.11.2020 06:50


Mathematics, 22.11.2020 06:50


Health, 22.11.2020 06:50


Biology, 22.11.2020 06:50

Social Studies, 22.11.2020 06:50