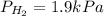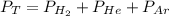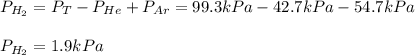Chemistry, 07.06.2020 01:00 montgomerykarloxc24x
The total pressure of a mixture of H2, He, and Ar is 99.3 kPa. The partial pressure of the He is 42.7 kPa, and the partial pressure of Ar is 54.7 kPa. What is the partial pressure of hydrogen

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 15:00
Answer explain why it is not possible to deduce a complete order of reactivity.
Answers: 3

Chemistry, 22.06.2019 20:00
Acm ruler with main graduations from 1 to 10 from left to right there are 10 secondary graduations between each of the main graduations there is a line that begins. at the left end of the ruler 10 secondary graduations to the left of the “1 main graduation the right end of the line ends on the eighth secondary graduation to the right of 3 how long is the line
Answers: 1

Chemistry, 22.06.2019 20:30
From the choices provided below, list the reagent(s) in order that will react with cyclopentanone to form the compound shown below.
Answers: 2
You know the right answer?
The total pressure of a mixture of H2, He, and Ar is 99.3 kPa. The partial pressure of the He is 42....
Questions

Mathematics, 30.10.2020 18:00

Physics, 30.10.2020 18:00


History, 30.10.2020 18:00

Health, 30.10.2020 18:00

Chemistry, 30.10.2020 18:00

Mathematics, 30.10.2020 18:00


Advanced Placement (AP), 30.10.2020 18:00



English, 30.10.2020 18:00


Mathematics, 30.10.2020 18:00


History, 30.10.2020 18:00


History, 30.10.2020 18:00