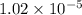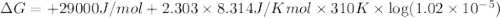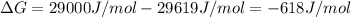Chemistry, 07.06.2020 05:00 21ghostrider21
The standard free energy change in physiological conditions (G') for the reaction catalyzed by malate dehydrogenase in the citric acid cycle malate + NAD+ oxaloacetate + NADH + H+ is ~+29 kJmol-1 Calculate the actual G' at 37C if Keq' is 1.02 × 10-5

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Electric charge is what ? a. kinetic energy b. radiation c. discovery d. electricity
Answers: 1

Chemistry, 22.06.2019 11:00
Which element would mostly likely have an electron affinity measuring closest to zero
Answers: 3

Chemistry, 22.06.2019 20:20
Which formula equation represents the burning of sulfur to produce sulfur dioxide? s(s) + o2(g) 4502(9) 2h2s(s) + 302(g) —> 2h20(0) + 2502(9) 4fes2+1102 —> 2fe2o3 + 8502 2802(g) + o2(9) v205 , 2503(9)
Answers: 1

Chemistry, 22.06.2019 21:30
In science class richard learns that a substance has a boiling point of 230 fahrenheit his teacher ask him to convert this temperature to degrees celsius what is the boiling point of his substance in degrees celsius
Answers: 3
You know the right answer?
The standard free energy change in physiological conditions (G') for the reaction catalyzed by malat...
Questions

Mathematics, 05.10.2020 15:01



Computers and Technology, 05.10.2020 15:01


Geography, 05.10.2020 15:01

Mathematics, 05.10.2020 15:01


Spanish, 05.10.2020 15:01

English, 05.10.2020 15:01

Mathematics, 05.10.2020 15:01

Mathematics, 05.10.2020 15:01

Biology, 05.10.2020 15:01

English, 05.10.2020 15:01



Mathematics, 05.10.2020 15:01



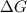 is -618 J/mol
is -618 J/mol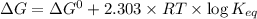
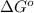 = standard free energy change = +29 kJ/mol =
= standard free energy change = +29 kJ/mol = 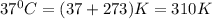
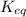 = equilibrium constant =
= equilibrium constant =