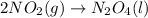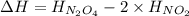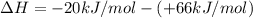Chemistry, 07.06.2020 13:57 Spence8900
PLZ HELP For the reaction: 2NO2(g) → N2O4(l),
the ΔH of the reactants, two moles of NO2 (g), is + 66 kJ/mol,
and the ΔH of the products, N2O4 (l), is -20 kJ/mol.
Which of the following shows the ΔH (change in enthalpy) for the reaction as a whole?
Question 8 options:
ΔrxnH =(- 20 kJ/mol) / (+66 kJ/mol)
ΔrxnH = (+66 kJ/mol) + (- 20 kJ/mol)
ΔrxnH =(-20 kJ/mol) - (+ 66 kJ/mol)
ΔrxnH =(+66 kJ/mol) - (- 20 kJ/mol)

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 14:00
The study of witch tree monkeys feed in is part of the science life
Answers: 1

Chemistry, 22.06.2019 03:20
What is the ima of the 1 st class lever in the graphic given? 2 3 0.5
Answers: 1

Chemistry, 22.06.2019 03:40
Chemical kinetics what was the rate of reaction in trial 3? choose the closest answer.
Answers: 3

Chemistry, 22.06.2019 12:00
A5.000 g sample of niso4 h2o decomposed to give 2.755 g of anhydrous niso4. what is the formula of the hydrate? what is the full chemical name for the hydrate? what is the molar mass of the hydrate? niso4•_h2o what is the mass % of water in the hydrate?
Answers: 1
You know the right answer?
PLZ HELP For the reaction: 2NO2(g) → N2O4(l),
the ΔH of the reactants, two moles of NO2 (g), is + 6...
Questions

Social Studies, 03.09.2020 23:01



Mathematics, 03.09.2020 23:01


English, 03.09.2020 23:01

Physics, 03.09.2020 23:01

Mathematics, 03.09.2020 23:01



Mathematics, 03.09.2020 23:01


Biology, 03.09.2020 23:01

History, 03.09.2020 23:01


English, 03.09.2020 23:01




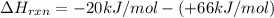
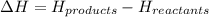
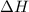 = enthalpy change = ?
= enthalpy change = ?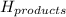 = enthalpy of products
= enthalpy of products 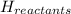 = enthalpy of reactants
= enthalpy of reactants