
Chemistry, 09.06.2020 03:57 tonimgreen17p6vqjq
The chemical formula of a compound describes the elements the compound contains and the
A. pH of the compound.
B. conduction of the compound.
C. ratio of elements in the compound.
D. ratio of neutrons to protons in each element.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1


Chemistry, 22.06.2019 21:30
How many liters of 3.0 m naoh solution will react with 0.60 liters of 4.0 m h2so4? h2so4 + naoh → na2so4 + h2o 1.2 l 1.6 l 2.4 l 2.8 l
Answers: 3
You know the right answer?
The chemical formula of a compound describes the elements the compound contains and the
A. pH of th...
Questions


Computers and Technology, 30.03.2021 04:20


Computers and Technology, 30.03.2021 04:20



Computers and Technology, 30.03.2021 04:20


Mathematics, 30.03.2021 04:20

Arts, 30.03.2021 04:20


Mathematics, 30.03.2021 04:20

Mathematics, 30.03.2021 04:20







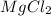 is the chemical formula for Magnesium chloride and it shows that the ratio of Magnesium to Chlorine (Cl) is 1:2.
is the chemical formula for Magnesium chloride and it shows that the ratio of Magnesium to Chlorine (Cl) is 1:2.

