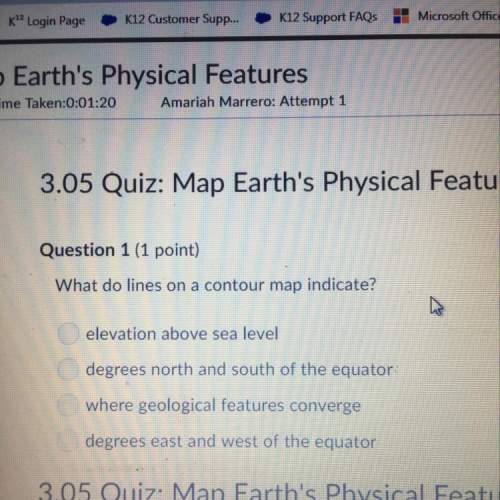
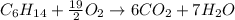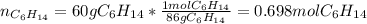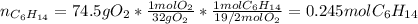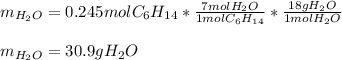Problem PageQuestion Liquid hexane CH3CH24CH3 will react with gaseous oxygen O2 to produce gaseous carbon dioxide CO2 and gaseous water H2O. Suppose 60. g of hexane is mixed with 74.5 g of oxygen. Calculate the maximum mass of water that could be produced by the chemical reaction. Round your answer to 3 significant digits.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 10:50
A100 kmol/h stream that is 97 mole% carbon tetrachloride (ccl4) and 3% carbon disulfide (cs2) is to be recovered from the bottom of a distillation column. the feed to the column is 16 mole% cs2 and 84% ccl4, and 2% of the ccl4 entering the column is contained in the overhead stream leaving the top of the column. calculate the mass and mole fractions of ccl4 in the overhead stream, and determine the molar flow rates of ccl4 and cs2 in the overhead and feed streams. 12. mw_ccla- 153.82; mw_cs2-76.14.
Answers: 3


Chemistry, 22.06.2019 16:50
What is conserved in the reaction shown below? h2(g) + cl2 (g) --> 2hcl(g)a. mass onlyb. mass and moles onlyc. mass, moles, and molecules onlyd. mass, moles, molecules, and volume
Answers: 2
You know the right answer?
Problem PageQuestion Liquid hexane CH3CH24CH3 will react with gaseous oxygen O2 to produce gaseous c...
Questions

Health, 19.01.2021 21:50




Chemistry, 19.01.2021 21:50

Chemistry, 19.01.2021 21:50



Mathematics, 19.01.2021 21:50

Mathematics, 19.01.2021 21:50

History, 19.01.2021 21:50







English, 19.01.2021 21:50


Mathematics, 19.01.2021 21:50