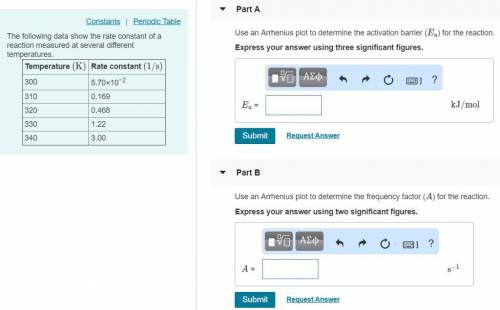
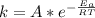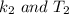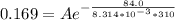The following data show the rate constant of a reaction measured at several different temperatures. Temperature (K) Rate Constant (1/s) 310 0.194 320 0.554 330 1.48 340 3.74 350 8.97 Part APart complete Use an Arrhenius plot to determine the activation barrier for the reaction. Express your answer using three significant figures. Ea

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:00
Noble gases are the most reactive elements on the periodic table. a. true b. false
Answers: 2

Chemistry, 22.06.2019 05:50
What are transitions between a liquid and gas called? identify which way they are transitioning
Answers: 2

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1

You know the right answer?
The following data show the rate constant of a reaction measured at several different temperatures....
Questions

Mathematics, 05.03.2021 06:50

Chemistry, 05.03.2021 06:50


Mathematics, 05.03.2021 06:50


History, 05.03.2021 06:50


History, 05.03.2021 06:50

History, 05.03.2021 06:50

Mathematics, 05.03.2021 06:50

English, 05.03.2021 06:50

Mathematics, 05.03.2021 06:50

Chemistry, 05.03.2021 06:50



History, 05.03.2021 06:50

Mathematics, 05.03.2021 06:50


English, 05.03.2021 06:50

Mathematics, 05.03.2021 06:50

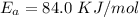

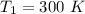
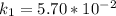
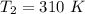
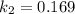
![ln [\frac{k_2}{k_1} ] = \frac{E_a}{R} [\frac{1}{T_1} - \frac{1}{T_2} ]](/tpl/images/0683/3945/2ceab.png)
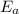 is the activation barrier for the reaction
is the activation barrier for the reaction 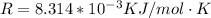
![ln [\frac{0.169}{6*10^-2{}} ] = \frac{E_a}{8.314*10^{-3}} [\frac{1}{300} - \frac{1}{310} ]](/tpl/images/0683/3945/6f5a8.png)