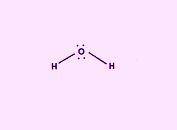
Draw the Lewis structure of H2O. Include any nonbonding electron pairs. Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons. - CHONSPFBrClIXMore Request Answer Part B What is the electron geometry of H2O

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:30
Asample of silver (with work function ? = 4.52 ev) is exposed to an ultraviolet light source (? = 200 nm), which results in the ejection of photoelectrons. what changes will be observed if: silver is replaced with copper (? = 5.10 ev) more photoelectrons ejected no photoelectrons are emitted fewer photoelectrons ejected more energetic photoelectrons (on average) less energetic photoelectrons (on average)
Answers: 3

Chemistry, 22.06.2019 12:40
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2


Chemistry, 23.06.2019 04:10
An unknown substance has been shown to have weak covalent bonds. which of the following is most likely a property of this substance? a. high ph b. high conductivity c. low melting point d. low flammability
Answers: 3
You know the right answer?
Draw the Lewis structure of H2O. Include any nonbonding electron pairs. Draw the molecule by placing...
Questions

Mathematics, 27.08.2019 06:20


Chemistry, 27.08.2019 06:20

Mathematics, 27.08.2019 06:20

Physics, 27.08.2019 06:20

Health, 27.08.2019 06:20

Biology, 27.08.2019 06:20

History, 27.08.2019 06:20

Mathematics, 27.08.2019 06:20



Health, 27.08.2019 06:20

Mathematics, 27.08.2019 06:20

Health, 27.08.2019 06:20

History, 27.08.2019 06:20

English, 27.08.2019 06:20

English, 27.08.2019 06:20

World Languages, 27.08.2019 06:20

Mathematics, 27.08.2019 06:20

 is, the total number of valence electrons is eight in
is, the total number of valence electrons is eight in