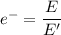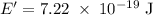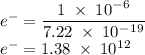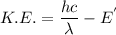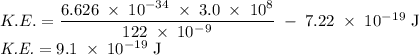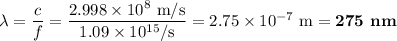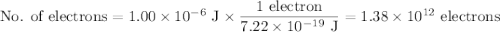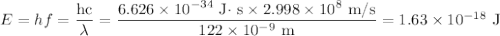Chemistry, 12.06.2020 05:57 coryowens44
Molybdenum metal requires a photon with a minimum frequency of 1.09x1015s-1before it can emit an electron via the photoelectric effect.
a) What is the minimum energy needed to eject an electron?
b)What wavelength of radiation (in nm) will provide a photon of this energy?
c)How many electrons can be freed by a burst of radiation whose total energy is 1.00 μJ, assuming one photon causes one electron to be freed? (μ= micro = 10-6)
d) If molybdenum is irradiated with light of 122nm, what is the maximum kinetic energy of the emitted electrons?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:00
An electron moved from shell n = 2 to shell n = 1. what most likely happened during the transition? a fraction of a photon was added. a photon of energy was absorbed. a fraction of a photon was removed. a photon of energy was released.
Answers: 1

Chemistry, 22.06.2019 14:30
100 grams of molten lead (600°c) is used to make musket balls. if the lead shot is allowed to cool to room temperature (21°c), what is the change in entropy (in j/k) of the lead? (for the specific heat of molten and solid lead use 1.29 j/g⋅°c; the latent heat of fusion and the melting point of lead are 2.45 × 104 j/kg and 327°c, respectively.)
Answers: 1


Chemistry, 22.06.2019 16:00
How do dying stars contribute to the formation of planets
Answers: 1
You know the right answer?
Molybdenum metal requires a photon with a minimum frequency of 1.09x1015s-1before it can emit an ele...
Questions

Chemistry, 05.05.2020 06:47

History, 05.05.2020 06:47




Social Studies, 05.05.2020 06:47



Biology, 05.05.2020 06:47


Mathematics, 05.05.2020 06:47

Mathematics, 05.05.2020 06:47

Geography, 05.05.2020 06:47



Social Studies, 05.05.2020 06:47


Physics, 05.05.2020 06:47


Chemistry, 05.05.2020 06:47

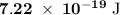 .
.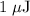 energy has been
energy has been 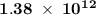 .
.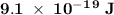 .
.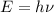
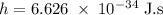
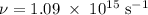
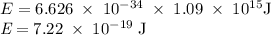
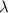 ) of the radiation has been given by:
) of the radiation has been given by: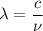
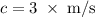
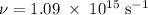 .
.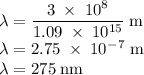
 ) burst out can be given as:
) burst out can be given as: