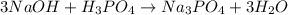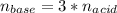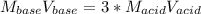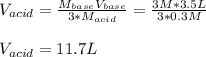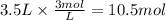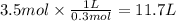Chemistry, 13.06.2020 19:57 mexicanvanilla
How many liters of 0.3m h3po4 are needed to neutralize 3.5l of 3m naoh

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Will mark brainliest26. which of these statements are true? (3 points)a. gases are compressibleb. gases fill their containers completelyc. the pressure of a gas is independent of the temperatured. gases have masse. gases exert pressuref. the pressure of a gas is dependent on the volumeg. gas pressure results from the collisions between gas particlesh. gases have a definite volume and shape
Answers: 1

Chemistry, 22.06.2019 04:00
The rules of engagement (roe) working group is often used to (select all that apply.)
Answers: 2

Chemistry, 22.06.2019 14:30
Aroom with dimensions 7.00m×8.00m×2.50m is to be filled with pure oxygen at 22.0∘c and 1.00 atm. the molar mass of oxygen is 32.0 g/mol. how many moles noxygen of oxygen are required to fill the room? what is the mass moxygen of this oxygen?
Answers: 1

Chemistry, 22.06.2019 20:00
Listenbase your answer to the question on the information below.nuclear radiation is harmful to living cells, particularly to fast-growing cells, such as cancer cells and blood cells. an external beam of the radiation emitted from a radioisotope can be directed on a small area of a person to destroy cancer cells within the body.cobalt-60 is an artificially produced radioisotope that emits gamma rays and beta particles. one hospital keeps a 100.0-gram sample of cobalt-60 in an appropriate, secure storage container for future cancer treatment.which choice represents the correct product for the beta decay of the co-60? fe-60ni-60fe-61ni-61
Answers: 2
You know the right answer?
How many liters of 0.3m h3po4 are needed to neutralize 3.5l of 3m naoh...
Questions


English, 05.07.2019 03:00

Mathematics, 05.07.2019 03:00

English, 05.07.2019 03:00


Mathematics, 05.07.2019 03:00


English, 05.07.2019 03:00

Arts, 05.07.2019 03:00

English, 05.07.2019 03:00


Mathematics, 05.07.2019 03:00

Social Studies, 05.07.2019 03:00

Geography, 05.07.2019 03:00

History, 05.07.2019 03:00



Mathematics, 05.07.2019 03:00