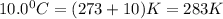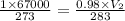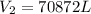Chemistry, 13.06.2020 23:57 marithatkid
if the balloon is 67,000 L at STP, what volume would it be if the air temperature cool to10.0°C in the pressure dropped to 99.2kPa as the balloon went higher?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:50
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1

Chemistry, 22.06.2019 03:30
The boiling point of liquids is very high what does it indicate
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 17:10
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2
You know the right answer?
if the balloon is 67,000 L at STP, what volume would it be if the air temperature cool to10.0°C in t...
Questions

Mathematics, 17.10.2019 01:00




Physics, 17.10.2019 01:00



History, 17.10.2019 01:00

Mathematics, 17.10.2019 01:00


History, 17.10.2019 01:00





Mathematics, 17.10.2019 01:00

Geography, 17.10.2019 01:00

Biology, 17.10.2019 01:00

History, 17.10.2019 01:00

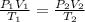
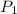 = initial pressure of gas (STP) = 1 atm
= initial pressure of gas (STP) = 1 atm 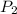 = final pressure of gas = 99.2 kPa = 0.98 atm (1kPa=0.0098atm)
= final pressure of gas = 99.2 kPa = 0.98 atm (1kPa=0.0098atm)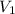 = initial volume of gas = 67000 L
= initial volume of gas = 67000 L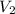 = final volume of gas = ?
= final volume of gas = ?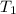 = initial temperature of gas (STP) = 273K
= initial temperature of gas (STP) = 273K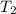 = final temperature of gas =
= final temperature of gas =