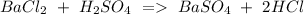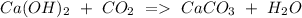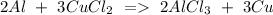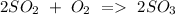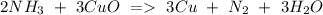Balance the following chemical equations.
a) Ba Cl2 + H2SO4 BaSO4 + HCl.
b) Calcium hy...

Chemistry, 13.06.2020 11:57 flyingcerberus1408
Balance the following chemical equations.
a) Ba Cl2 + H2SO4 BaSO4 + HCl.
b) Calcium hydroxide + Carbon dioxide Calcium carbonate + Water.
c) Aluminum + Copper chloride Copper + Aluminum chloride
d) Sulphur dioxide + Oxygen Sulphur trioxide
e) NH3+ CuO Cu + N2 + H2O

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 21:30
The solid xy decomposes into gaseous x and y: xy(s) m x(g) + y(g) kp = 4.1 (at 0 °c) if the reaction is carried out in a 22.4 l container, which initial amounts of x and y will result in the formation of solid xy?
Answers: 1
You know the right answer?
Questions





Advanced Placement (AP), 01.11.2019 23:31

English, 01.11.2019 23:31

Chemistry, 01.11.2019 23:31

History, 01.11.2019 23:31

Mathematics, 01.11.2019 23:31


English, 01.11.2019 23:31


Health, 01.11.2019 23:31


Mathematics, 01.11.2019 23:31


Mathematics, 01.11.2019 23:31


Mathematics, 01.11.2019 23:31

Biology, 01.11.2019 23:31