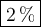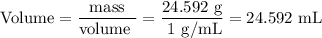Chemistry, 16.06.2020 01:57 elisakgarcia2007
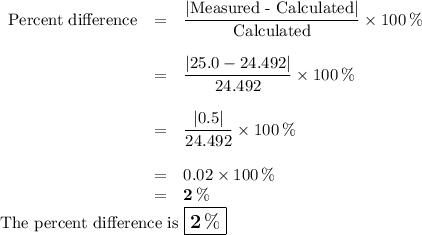
DATA AND CALCULATIONS: (you must show your calculations) Part I. Determination of accuracy of a graduated cylinder Calculations: Experimental Step Measurable Mass of empty graduated cylinder 47.229 g Mass of filled graduated cylinder 71.821 g Mass of water (filled – empty) g Volume of water, calculated (calculated from mass of water, using the equation “density = mass/volume”, given the fact that the density of water is exactly 1 g/mL) mL Volume of water, measured (from the reading of the scale on the graduated cylinder) 25.0 mL Percent difference between measured and calculated volumes of water [(measured-calculated)/calculated] ×100% %

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
How many moles are in 250 grams of tungsten (w)? * 4.4x10^23 moles 4.2x10^23 moles 0.7 moles 1.4 moles
Answers: 3

Chemistry, 22.06.2019 08:00
Joe shines white light into a bowl half full of water at an angle of incident of 27.5°. calculate the angle of refraction in the water given the indices of refraction for air and water are 1.00 and 1.36, respectively.
Answers: 2

Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1

Chemistry, 22.06.2019 19:20
The equation picture below shows which type of nuclear reaction u 235 + n x e 134 + sr 100 + 2n
Answers: 1
You know the right answer?
DATA AND CALCULATIONS: (you must show your calculations) Part I. Determination of accuracy of a grad...
Questions

Spanish, 01.09.2019 05:10

Mathematics, 01.09.2019 05:10

English, 01.09.2019 05:10


Mathematics, 01.09.2019 05:10

History, 01.09.2019 05:10

English, 01.09.2019 05:10


English, 01.09.2019 05:10





History, 01.09.2019 05:10




History, 01.09.2019 05:10

Mathematics, 01.09.2019 05:10

History, 01.09.2019 05:10