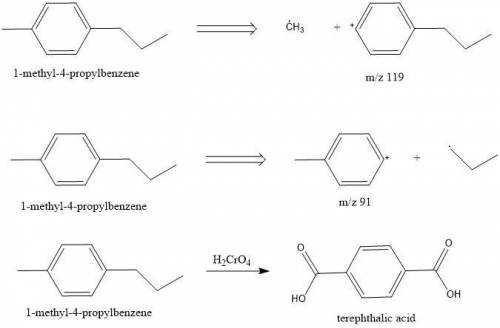
Compounds A and B (both C10H14) show prominent peaks in their mass spectrum at m/z 134 and 119. Compound B also shows a less prominent peak at m/z 91. On vigorous oxidation with chromic acid, compound A is nonreactive while compound B yielded terephthalic acid.
From this information, deduce the structures of both compounds, and then draw the structure of B.
You do not have to consider stereochemistry
You do not have to explicitly draw H atoms

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:20
What are the spectator ions in 2h+ + so42- + ca2+ + 2r → caso4 + 2h+ + 21?
Answers: 1


Chemistry, 23.06.2019 01:00
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2

Chemistry, 23.06.2019 05:00
Match the term to its description match term definition chemical energy a) internal energy caused by vibrations of atoms and molecules electrical energy b) electromagnetic energy that travels in waves radiant energy c) the movement of an electrical charge thermal energy d) potential energy stored in the bonds between atoms
Answers: 1
You know the right answer?
Compounds A and B (both C10H14) show prominent peaks in their mass spectrum at m/z 134 and 119. Comp...
Questions






Mathematics, 18.06.2020 18:57






History, 18.06.2020 18:57

English, 18.06.2020 18:57




Computers and Technology, 18.06.2020 18:57

Mathematics, 18.06.2020 18:57