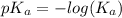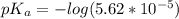Chemistry, 16.06.2020 19:57 Rachaeltice8810
Since some amino acids, such as Glu and His, can be viewed as weak acids or bases, the ratio of their protonated form to their deprotonated form is dependent on pH. Furthermore, you can use this knowledge to predict whether a given amino acid will exist predominantly in its protonated or deprotonated form at a given pH. To do this, it is easier to think about the pK, of the amino acid functional group. Recall from general chemistry that the pK, is equal to -log(K). Using the K, value for the Glu side chain, which is 5.62 x 10-5, calculate the pK, for the Glu side chain. pKa =

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
The continuous release of nuclear energy caused when one fission reaction triggered more nuclear reactions is a
Answers: 3

Chemistry, 22.06.2019 04:50
The name of the ion, s2-, is: sulfurous ion sulfide ion sulfur ion sulfate ion
Answers: 1


Chemistry, 22.06.2019 11:30
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2
You know the right answer?
Since some amino acids, such as Glu and His, can be viewed as weak acids or bases, the ratio of thei...
Questions






Geography, 28.08.2019 11:30

Mathematics, 28.08.2019 11:30


Biology, 28.08.2019 11:30




Computers and Technology, 28.08.2019 11:30


Mathematics, 28.08.2019 11:30


Social Studies, 28.08.2019 11:30


Geography, 28.08.2019 11:30

Biology, 28.08.2019 11:30

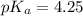
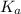 value of Glu is
value of Glu is 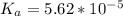
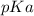 is mathematically represented as
is mathematically represented as