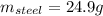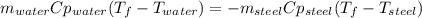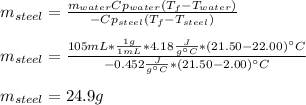Chemistry, 18.06.2020 02:57 Jessietorres2123
A volume of 105 mL of H2O is initially at room temperature (22.00 ∘C). A chilled steel rod at 2.00 ∘C is placed in the water. If the final temperature of the system is 21.50 ∘C , what is the mass of the steel bar? Use the following values: specific heat of water = 4.18 J/(g⋅∘C) specific heat of steel = 0.452 J/(g⋅∘C) Express your answer to three significant figures and include the appropriate units.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:00
Explain how the integumentary system plays a crucial role in the ability to maintain homeoestasis
Answers: 1


Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 1

Chemistry, 22.06.2019 21:00
Which of the following is a physical property flammability heat of combustion solubility and toxicity
Answers: 1
You know the right answer?
A volume of 105 mL of H2O is initially at room temperature (22.00 ∘C). A chilled steel rod at 2.00 ∘...
Questions


Geography, 28.01.2020 15:57


Biology, 28.01.2020 15:57

Geography, 28.01.2020 15:57

History, 28.01.2020 15:57


History, 28.01.2020 15:57



Geography, 28.01.2020 15:57


Mathematics, 28.01.2020 15:57


Arts, 28.01.2020 15:57

English, 28.01.2020 15:57



Health, 28.01.2020 15:57