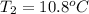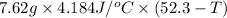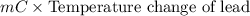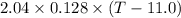Chemistry, 19.06.2020 00:57 straightbarz5916
A 2.04 g lead weight, initially at 10.8 oC, is submerged in 7.62 g of water at 52.3 oC in an insulated container. clear = 0.128 J/g oF; water = 4.18 J/goC. What is the final temperature of both the weight and the water at thermal equilibrium

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Now consider the reaction when 45.0 g naoh have been added. what amount of naoh is this, and what amount of fecl3 can be consumed by it?
Answers: 3

Chemistry, 21.06.2019 22:40
How many electrons does silver have to give up in order to achieve a sido noble gas electron configuration
Answers: 1

Chemistry, 21.06.2019 23:50
2points why do scientists need governmental funding? o a. government politicians ask all the important scientific questions. o b. scientists have to pay taxes to the government on the money they make. o c. the cost of doing scientific research can be very high. o d. the government is controlled by scientists. submit
Answers: 3

You know the right answer?
A 2.04 g lead weight, initially at 10.8 oC, is submerged in 7.62 g of water at 52.3 oC in an insulat...
Questions

Geography, 18.08.2019 13:30

Mathematics, 18.08.2019 13:30

Biology, 18.08.2019 13:30




Mathematics, 18.08.2019 13:30



History, 18.08.2019 13:30

Mathematics, 18.08.2019 13:30





Geography, 18.08.2019 13:30

Mathematics, 18.08.2019 13:30


History, 18.08.2019 13:30

Mathematics, 18.08.2019 13:30

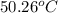 .
.