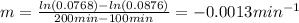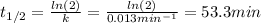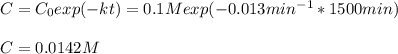3.The following data of decomposition reaction of thionyl chloride (SO2Cl2) were collected at a certain temperature and the concentration of SO2Cl2 were monitored as shown in the table.
SO2Cl2 (g) SO2 (g) + Cl2 (g)
Time (min)Conc. of SO2Cl2 (mol/L)
00.1000
1000.0876
2000.0768
3000.0673
4000.0590
5000.0517
6000.0453
7000.0397
8000.0348
9000.0305
10000.0267
11000.0234
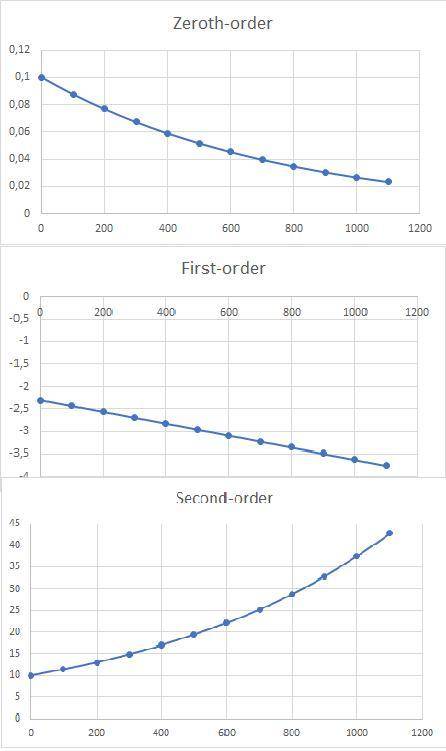
a)Determine graphically whether the kinetics of the reaction is zero order, first order or second order with respect to SO2Cl2 and then write the rate equation.
b)Determine the rate constant (k) of the reaction.
c)Determine the half-life (t½) for the reaction.
d)What will be the concentration of SO2Cl2 left in the reaction mixture at 1500 minutes?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which type of orbits are found in the principal energy level n = 2 a - s b - s, f c - s, d d - s, p e - s, p, d
Answers: 1


Chemistry, 22.06.2019 16:40
Identify the lewis acid in this balanced equation: ag+ + 2nh3 -> ag(nh3)2+a. ag+b. nh3c. ag(nh3)2+
Answers: 1

Chemistry, 23.06.2019 00:10
Find the missing probability in the table below a.0.10 b.40 c.0.80 d. 0.20
Answers: 2
You know the right answer?
3.The following data of decomposition reaction of thionyl chloride (SO2Cl2) were collected at a cert...
Questions





Spanish, 07.02.2022 22:30

History, 07.02.2022 22:30


Mathematics, 07.02.2022 22:30



English, 07.02.2022 22:30


History, 07.02.2022 22:30

Mathematics, 07.02.2022 22:30



Mathematics, 07.02.2022 22:30

Mathematics, 07.02.2022 22:30