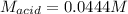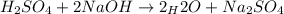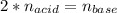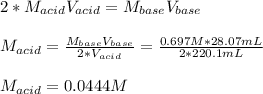Suppose you are titrating a sulfuric acid solution of unknown concentration with a sodium hydroxide solution according to the equation H 2 S O 4 + 2 N a O H ⟶ 2 H 2 O + N a 2 S O 4 If you require 28.07 mL of 0.697 M NaOH solution to titrate 220.1 mL of H 2 SO 4 solution, what is the concentration of the H 2 SO 4 solution? Type

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 21:30
Athe top of a hill, an athlete on a skateboard has x joules of mechanical energy. how much mechanical energy will she have at the bottom of the hill? ignore the effects of friction.
Answers: 1

Chemistry, 23.06.2019 01:00
What type of chemical bond is formed between two atoms of bromine 1. metallic 2. hydrogen 3. ionic 4. covalent
Answers: 1


Chemistry, 23.06.2019 13:30
Explain the impact that changing the temperature has on a system in a state of dynamic equilibrium. what will happen when the temperature of an exothermic reaction mixture at equilibrium is increased?
Answers: 3
You know the right answer?
Suppose you are titrating a sulfuric acid solution of unknown concentration with a sodium hydroxide...
Questions

Mathematics, 28.08.2019 13:10



Mathematics, 28.08.2019 13:10

Biology, 28.08.2019 13:10

Mathematics, 28.08.2019 13:10

Mathematics, 28.08.2019 13:10

Mathematics, 28.08.2019 13:10




History, 28.08.2019 13:10



Chemistry, 28.08.2019 13:10

Business, 28.08.2019 13:10

Chemistry, 28.08.2019 13:10



Mathematics, 28.08.2019 13:10