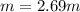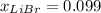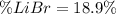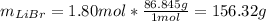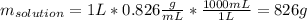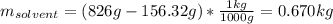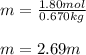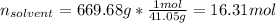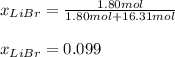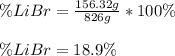Chemistry, 20.06.2020 22:57 shaylawaldo11
Acetonitrile, CH3CN, is a polar organic solvent that dissolves many solutes, including many salts. The density of a 1.80 M acetonitrile solution of LiBr is 0.826 g/mL. Calculate the concentration of the solution in units of (a) molality; (b) mole fraction of LiBr; (c) mass percentage of CH3CN.

Answers: 3
Another question on Chemistry


Chemistry, 21.06.2019 22:30
Check the correct box to describe the periodic trends in electronegativity. electronegativity across a period: decreases. increases. electronegativity down a group: decreases. increases.
Answers: 2


Chemistry, 22.06.2019 04:00
Drag each label to the correct location on the chart. classify each reaction as endothermic or exothermic.
Answers: 1
You know the right answer?
Acetonitrile, CH3CN, is a polar organic solvent that dissolves many solutes, including many salts. T...
Questions


Mathematics, 03.02.2020 15:05

Mathematics, 03.02.2020 15:05



Chemistry, 03.02.2020 15:05

English, 03.02.2020 15:05

Mathematics, 03.02.2020 15:05

Physics, 03.02.2020 15:05


English, 03.02.2020 15:05




Social Studies, 03.02.2020 15:05

Social Studies, 03.02.2020 15:05


Geography, 03.02.2020 15:05