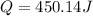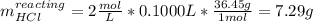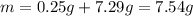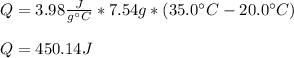Chemistry, 23.06.2020 10:57 brownvester44
4.The reaction of 0.25 g magnesium with 100.0 mL of 2 M hydrochloric acid (density d = 1.039 g/ml) raises the temperature of the solution from 20.0 °C to 35.0 °C. The specific heat for HCl is 3.98 J/(g. C). Mg(s) + 2HCl(aq) —> MgCl2(aq) + H2(g) Calculate the heat of the reaction. (Use the equation Q = s . m .ΔT where s is the specific heat, m is the mass of solution and ΔT is the change in temperature)

Answers: 2
Another question on Chemistry


Chemistry, 21.06.2019 21:30
If i make a solution by adding 83grams of sodium hydroxide to 750ml i’d water what is the molarity of sodium hydroxide
Answers: 1

Chemistry, 22.06.2019 00:30
Maria wants to determine which type of disinfectant kills the most bacteria. which of the following is the best way for maria to determine this? a. ask ten different companies that make disinfectants which type is best. b. put the same amount and species of bacteria on ten identical plates, and add ten different kinds of disinfectant to each plate. c. interview ten different people to determine which type of disinfectant they prefer. d. put the same amount and species of bacteria on ten identical plates, and add a different disinfectant to each plate.
Answers: 1

You know the right answer?
4.The reaction of 0.25 g magnesium with 100.0 mL of 2 M hydrochloric acid (density d = 1.039 g/ml) r...
Questions

Mathematics, 24.09.2020 21:01


Mathematics, 24.09.2020 21:01



Mathematics, 24.09.2020 21:01

Geography, 24.09.2020 21:01






English, 24.09.2020 21:01

Chemistry, 24.09.2020 21:01



Computers and Technology, 24.09.2020 21:01


Spanish, 24.09.2020 21:01