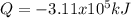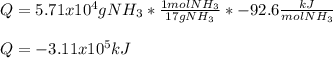Chemistry, 26.06.2020 16:01 soliseric879
Determine the amount of heat (in kJ) associated with the production of 5.71 × 104 g of ammonia according to the following equation. N2(g) + 3H2(g) 2NH3ΔH°rxn = −92.6 kJ Assume that the reaction takes place under standard-state conditions at 25°C.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1

Chemistry, 22.06.2019 21:00
How many neutrons does an element have if its atomic number is 50 and its mass number is 166
Answers: 1

Chemistry, 22.06.2019 22:30
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1

Chemistry, 22.06.2019 23:00
What is the measured amount of a product obtained from a chemical reaction?
Answers: 1
You know the right answer?
Determine the amount of heat (in kJ) associated with the production of 5.71 × 104 g of ammonia accor...
Questions

Social Studies, 28.08.2019 15:50

English, 28.08.2019 15:50


English, 28.08.2019 15:50


Advanced Placement (AP), 28.08.2019 15:50

English, 28.08.2019 15:50

Biology, 28.08.2019 15:50

Business, 28.08.2019 15:50





English, 28.08.2019 15:50

History, 28.08.2019 15:50





Social Studies, 28.08.2019 15:50