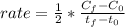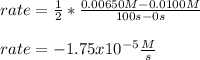Chemistry, 26.06.2020 16:01 emilyswinge4421
Nitrogen dioxide decomposes to nitric oxide and oxygen via the reaction: 2NO2 → 2NO + O2 In a particular experiment at 300 °C, [NO2] drops from 0.0100 to 0.00650 M in 100 s. The rate of disappearance of NO2 for this period is .

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1


Chemistry, 22.06.2019 21:30
What is happening when the water inside a kettle heats up and begins to boil
Answers: 1

Chemistry, 22.06.2019 23:30
The ammonia molecule in the diagram has the observed bond orientation because
Answers: 1
You know the right answer?
Nitrogen dioxide decomposes to nitric oxide and oxygen via the reaction: 2NO2 → 2NO + O2 In a partic...
Questions


History, 23.11.2020 18:30


Mathematics, 23.11.2020 18:30


Mathematics, 23.11.2020 18:30


Spanish, 23.11.2020 18:30



World Languages, 23.11.2020 18:30

Mathematics, 23.11.2020 18:30


English, 23.11.2020 18:30





English, 23.11.2020 18:30

Mathematics, 23.11.2020 18:30