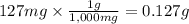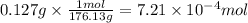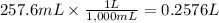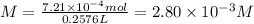Chemistry, 01.07.2020 16:01 Roninsongrant
Given that one cup = 257.6 mL, calculate the molarity of vitamin C in orange juice. Express your answer to an appropriate number of significant figures with the appropriate units.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:00
Nickel crystallizes in the face-centered cubic (fcc) lattice. the density of the metal is 8902 kg/m3. calculate the radius of a nickel atom.
Answers: 1

Chemistry, 22.06.2019 03:30
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 23.06.2019 00:30
What is the percent by mass of magnesium sulfate in mgso4.7h2o
Answers: 3
You know the right answer?
Given that one cup = 257.6 mL, calculate the molarity of vitamin C in orange juice.
Express your an...
Questions





Biology, 19.07.2019 10:10


Biology, 19.07.2019 10:10



Computers and Technology, 19.07.2019 10:10

Spanish, 19.07.2019 10:10



Mathematics, 19.07.2019 10:10

English, 19.07.2019 10:10

History, 19.07.2019 10:10

Physics, 19.07.2019 10:10