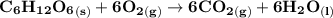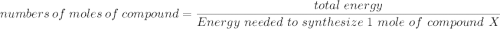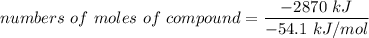Chemistry, 02.07.2020 22:01 treypickich14
Assume that the complete combustion of one mole of glucose, a monosaccharide, to carbon dioxide and water liberates 2870 kJ2870 kJ of energy (ΔG°′=−2870 kJ/mol(ΔG°′=−2870 kJ/mol ). If the energy generated by the combustion of glucose is entirely converted to the synthesis of a hypothetical compound X, calculate the number of moles of the compound that could theoretically be generated. Use the value ΔG°′compound X=−54.1 kJ/molΔG°′compound X=−54.1 kJ/mol . Round your answer to two significant figures.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 08:00
Match the mixture with the substance// i really need on this guys (it’s a pic btw)
Answers: 1

Chemistry, 22.06.2019 09:30
1. explain hydrogen peroxide, h 2 o 2 properties and decomposition reaction. 2. describe how each of the following natural cycles plays a part in earth’s climate system. (a) the water cycle (b) the carbon cycle
Answers: 1

You know the right answer?
Assume that the complete combustion of one mole of glucose, a monosaccharide, to carbon dioxide and...
Questions















History, 27.09.2019 05:10

History, 27.09.2019 05:10


Mathematics, 27.09.2019 05:10


 53 mole
53 mole