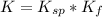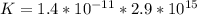Chemistry, 02.07.2020 16:01 jalynholden07
Consider the insoluble compound zinc carbonate , ZnCO3 . The zinc ion also forms a complex with hydroxide ions . Write a balanced net ionic equation to show why the solubility of ZnCO3 (s) increases in the presence of hydroxide ions and calculate the equilibrium constant for this reaction. For Zn(OH)42- , Kf = 2.9×1015 . Use the pull-down boxes to specify states such as (aq) or (s).

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 16:10
Given the following equation: 2a1 + 3mgcl2 --> 2alcl3 + 3mg how many moles of aluminum chloride are produced from 2.5 moles of magnesium chloride?
Answers: 1

Chemistry, 23.06.2019 01:00
Who examines and coordinates the cleanup of polluted sites?
Answers: 2

You know the right answer?
Consider the insoluble compound zinc carbonate , ZnCO3 . The zinc ion also forms a complex with hydr...
Questions

Mathematics, 12.05.2021 04:20

Mathematics, 12.05.2021 04:20

History, 12.05.2021 04:20







Mathematics, 12.05.2021 04:20


Mathematics, 12.05.2021 04:20

Mathematics, 12.05.2021 04:20

Health, 12.05.2021 04:20

Mathematics, 12.05.2021 04:20

Mathematics, 12.05.2021 04:20

Mathematics, 12.05.2021 04:20

Mathematics, 12.05.2021 04:20

Mathematics, 12.05.2021 04:20

![ZnCO_3 _{(s)} + 4 OH^{-}_{(aq)} \to [Zn(OH)_4]^{2-} _{(aq)} + CO_3^{2-} _{(aq)}](/tpl/images/0699/8948/a1741.png)
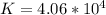
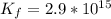
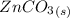 ⇔
⇔ 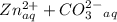 The solubility product constant for stage is
The solubility product constant for stage is 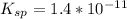
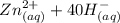 ⇔
⇔ ![[Zn(OH_4)]^{2-} _{(aq)}](/tpl/images/0699/8948/a852d.png) The formation constant for this step is given as
The formation constant for this step is given as