
Chemistry, 02.07.2020 17:01 emmaja121003
The value of ΔG°′ΔG°′ for the conversion of glucose-6-phosphate to fructose-6-phosphate (F6P) is +1.67 kJ/mol+1.67 kJ/mol . If the concentration of glucose-6-phosphate at equilibrium is 2.65 mM2.65 mM , what is the concentration of fructose-6-phosphate? Assume a temperature of 25.0°C25.0°C .

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be
Answers: 1


Chemistry, 22.06.2019 21:30
Describe at least two advantages and two disadvantages of using hydropower as a source of energy.
Answers: 2

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
The value of ΔG°′ΔG°′ for the conversion of glucose-6-phosphate to fructose-6-phosphate (F6P) is +1....
Questions



History, 21.09.2019 18:50

Health, 21.09.2019 18:50

Biology, 21.09.2019 18:50

Social Studies, 21.09.2019 18:50

Geography, 21.09.2019 18:50

Mathematics, 21.09.2019 18:50

Mathematics, 21.09.2019 18:50

History, 21.09.2019 18:50

Mathematics, 21.09.2019 18:50

Chemistry, 21.09.2019 18:50


Mathematics, 21.09.2019 18:50



Mathematics, 21.09.2019 18:50


History, 21.09.2019 18:50

Mathematics, 21.09.2019 18:50

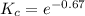
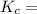 0.511
0.511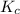
![K_c = \dfrac{[F6P]}{[G6P]}](/tpl/images/0699/9197/8414f.png)
![0.511 = \dfrac{[F6P]}{[2.65]}](/tpl/images/0699/9197/b9eb4.png)


