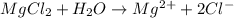In the compound mgcl2, the subscript 2 indicates that
a. there are two magnesium ions for each...

Chemistry, 23.08.2019 05:30 victoriakraus3497
In the compound mgcl2, the subscript 2 indicates that
a. there are two magnesium ions for each ion of chlorine
b. the chloride ion is twice the size of the magnesium ion.
c. magnesium and chlorine form a double covalent bond.
d. there are two chloride ions for each magnesium ion.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 08:30
Which of the following would have less momentum than a 52 kg cheetah running at 10 m/s?
Answers: 2

Chemistry, 22.06.2019 14:00
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1

Chemistry, 23.06.2019 01:30
Which statement justifies that hydrogen peroxide (h2o2) is a polar molecule? the o – h bond is nonpolar and the molecule is asymmetric. the o – h bond is nonpolar and the molecule is symmetric. the o – h bond is polar and the molecule is asymmetric. the o – h bond is polar and the molecule is symmetric.
Answers: 1
You know the right answer?
Questions

Mathematics, 20.04.2021 03:20





English, 20.04.2021 03:20


Mathematics, 20.04.2021 03:20


Mathematics, 20.04.2021 03:20

Chemistry, 20.04.2021 03:20


Social Studies, 20.04.2021 03:20

Mathematics, 20.04.2021 03:20






History, 20.04.2021 03:20

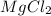 .
.