

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:10
How many moles of gas are present if p=11 atm, v=12l, t=185k?
Answers: 1

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1

Chemistry, 22.06.2019 14:30
How do temperature and salinity affect deepwater currents? as temperatures and salinity levels of water increase, the water rises to the surface where it creates currents as it moves to colder regions. they create changes in wind direction, moving denser water in the same direction as the wind and causing the deepwater circulation patterns found in the ocean. they equalize the forces on undersea currents caused by the coriolis effect as they replace more dense water with less dense water. they create density differences that cause dense deepwater currents to flow toward the equator where they displace less dense, warmer water above them.
Answers: 2

Chemistry, 23.06.2019 09:20
La reaccion entre monoxido de nitrogeno (no) y oxigeno para formardioxido de nitrogeno (no2) es un paso determinante para la formacion del smog, la reaccion es la siguiente: 2no + o2 = 2no2 cual sera el numero de moles de no2 que se formaran por la reaccion completa de 8 moles de oxigeno con suficiente monoxido?
Answers: 1
You know the right answer?
A titration of 200.0 mL of 1.00 M H2A was done with 1.38 M NaOH. For the diprotic acid H2A, Ka1 = 2....
Questions



Mathematics, 30.01.2020 03:04



Mathematics, 30.01.2020 03:04


Physics, 30.01.2020 03:04

Mathematics, 30.01.2020 03:04




History, 30.01.2020 03:04


History, 30.01.2020 03:04


English, 30.01.2020 03:04


History, 30.01.2020 03:04

English, 30.01.2020 03:04

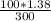 = 0.46
= 0.46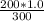 = 0.667
= 0.667
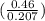 = 4.95
= 4.95 

