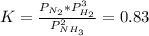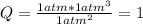Chemistry, 07.07.2020 21:01 kingtrent81
For the reaction below, initially the partial pressure of all 3 gases is 1.0atm. . 2NH3(g)--> N2(g) + 3H2(g) K, 0.83 1. When the reaction reach equilibrium the partial pressure of N2 will be greater than 1atm The reaction would shift toward the reactants The reaction would shift toward the products 2. When the reaction reach equilibrium the partial pressure of NH3 will be greater than 1atm 3. When the reaction reach equilibrium the partial pressure of H2 will be greater than 1atm

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 05:30
What happens to the atomic radius when an elctron is lost
Answers: 1

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 15:20
Select the most likely product for this reaction: koh(aq) + co2(g) – ? k2co3(aq) + h2o(1) k(s) + h2(g) + o2(g) k(s) + co3(9) +h2
Answers: 2
You know the right answer?
For the reaction below, initially the partial pressure of all 3 gases is 1.0atm. . 2NH3(g)--> N2(...
Questions

Advanced Placement (AP), 18.03.2021 01:20

Arts, 18.03.2021 01:20





Mathematics, 18.03.2021 01:20













Mathematics, 18.03.2021 01:20