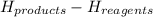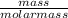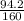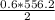A chemist measures the energy change Delta H during the following
2Fe2O3(s)->4FeO(s)+O2(g).
1) this reactions is: Endothermic or exothermic.
2) suppose 94.2g of Fe2O3 react. will any heat be relased or absorbed. yes absorbed. yes releases. no.
3) If you said heat will be released or absorbed in the second part of the question. calculate how much heat will be absored or released. be sure your answer has correct number of significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 22.06.2019 12:30
Which element has the lowest electronegativity? calcium(ca) gallium(ga) selenium(se) bromine(br)
Answers: 1

Chemistry, 22.06.2019 18:30
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1

Chemistry, 22.06.2019 21:00
One similarity and one difference between an element and a mixture of elements
Answers: 1
You know the right answer?
A chemist measures the energy change Delta H during the following
2Fe2O3(s)->4FeO(s)+O2(g).
Questions



Mathematics, 27.05.2021 23:00

Mathematics, 27.05.2021 23:00

Mathematics, 27.05.2021 23:00


Mathematics, 27.05.2021 23:00

Mathematics, 27.05.2021 23:00

Mathematics, 27.05.2021 23:00

Mathematics, 27.05.2021 23:00

Mathematics, 27.05.2021 23:00



Mathematics, 27.05.2021 23:00



Mathematics, 27.05.2021 23:00