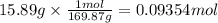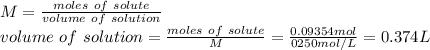A student wants to make a 0.250 M aqueous solution of silver nitrate, AgNO3, and has a bottle containing 15.89 g of silver nitrate. What should be the final volume of the solution? When you give your numerical answer, what is the correct significant figures and how do you know that is the correct amount?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:10
The covalent compound acetylene, which is the fuel of the oxyacetylene torch used by welders, has the molecular formula c2h2. the covalent compound benzene, a commercial solvent, has the molecular formula c6h6 each of these covalent compounds contains carbon and hydrogen atoms in a one-to-one ratio. would it be correct to write the chemical formulas of each as ch? explain.
Answers: 1

Chemistry, 22.06.2019 07:30
What is i fracture in the crust called when land move up, down or sideways
Answers: 2

Chemistry, 22.06.2019 13:30
1) which of the following is the best example of a physical change? a) sugar dissolving in tea b) firefly glowing 2) in the combustion of ethane, what is/are the reactants? c2h6 + o2 ==> co2 + h2o a) c2h6 and o2 b) co2 and c2h6
Answers: 2

Chemistry, 23.06.2019 00:30
Element j is 1s 2s 2p 3s . (i) how many unpaired electrons does j have? (ii) is j a good oxidizing agent or a reducing agent? (iii) state reason for the answer.
Answers: 1
You know the right answer?
A student wants to make a 0.250 M aqueous solution of silver nitrate, AgNO3, and has a bottle contai...
Questions



Mathematics, 28.04.2021 21:50

Chemistry, 28.04.2021 21:50


History, 28.04.2021 21:50

English, 28.04.2021 21:50


Mathematics, 28.04.2021 21:50

Social Studies, 28.04.2021 21:50

English, 28.04.2021 21:50




Physics, 28.04.2021 21:50



Mathematics, 28.04.2021 21:50