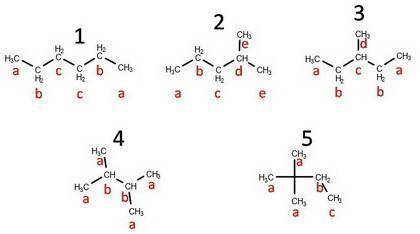
There are five constitutional isomers with the molecular formula C6H14. When treated with chlorine at 300°C, isomer A gives a mixture of two monochlorination products. Under the same conditions, isomer B gives a mixture of five monochlorination products, isomer C gives four monochlorination products, and isomer D gives a mixture of three monochlorination products. From this information, draw the structural formula of isomer D.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Reaction rate depends on how many molecules are coming into contact with each other with enough energy to react. increasing the temperature of the reactants will increase -
Answers: 3

Chemistry, 22.06.2019 17:00
In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the final internal energy of the system if the inital energy was 2000 j we have to do all of these down here 1)write the equation 2)list out your know variables 3)plug the numbers into the equations 4)solve 5)write your solution statemtn that includes inital energuy and final energuy added
Answers: 1

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium chlorate
Answers: 3

You know the right answer?
There are five constitutional isomers with the molecular formula C6H14. When treated with chlorine a...
Questions


Mathematics, 09.03.2021 19:30

English, 09.03.2021 19:30



Chemistry, 09.03.2021 19:30

Mathematics, 09.03.2021 19:30

Arts, 09.03.2021 19:30

Mathematics, 09.03.2021 19:30


History, 09.03.2021 19:30

English, 09.03.2021 19:30

Mathematics, 09.03.2021 19:30





Mathematics, 09.03.2021 19:30