Chemistry, 14.07.2020 20:01 gizmo50245
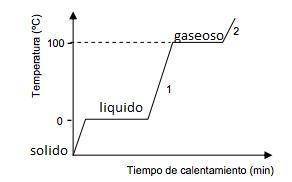
Describe what you would see if you graphed the heating curve for water, going from ice to gas. Why is a heating curve not a straight line? Include temperatures of phase changes and describe the slopes of the line through various temperature regions. You may draw a diagram if you are writing your response by hand.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:30
For each of the following mixtures decide if filtering would be suitable to separate the substances. explain your answers. oil in water sugar in water sand in water chalk in water tea leaves in a cup of tea
Answers: 2

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2

Chemistry, 22.06.2019 22:00
All of the following are homogeneous mixtures except a) sugar dissolved in water. b) orange juice. c) coffee with cream. d) household vinegar. e) apple juice
Answers: 1

Chemistry, 23.06.2019 00:30
You are attempting to recrystallize a crude product mixture. you add the appropriate amount of hot solvent and are allowing the solution to slowly cool to room temperature. however, at room temperature no crystals have appeared, which of the following methods should be used to induce crystallization? choose all correct answers. a) place the flask in an ice bath. b) swirl the contents of the flask. c) add a small seed crystal of the desired product. d) scratch the inside of the glassware using a stir rod. it can be multiple choices
Answers: 3
You know the right answer?
Describe what you would see if you graphed the heating curve for water, going from ice to gas. Why i...
Questions

Mathematics, 15.12.2019 20:31

English, 15.12.2019 20:31



History, 15.12.2019 20:31


Mathematics, 15.12.2019 20:31

Mathematics, 15.12.2019 20:31


World Languages, 15.12.2019 20:31

Physics, 15.12.2019 20:31

SAT, 15.12.2019 20:31


Chemistry, 15.12.2019 20:31


Mathematics, 15.12.2019 20:31

History, 15.12.2019 20:31