
Chemistry, 14.07.2020 02:01 hubbabubba0715
Calculate the pH of a 0.0255 M solution of ammonium bromide (NH4Br). The Kb of ammonia is 1.76 x 10-5

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:00
The diagram below shows a cell placed in a solution.a cell is shown placed inside a beaker. it is labeled cell. the solution inside the beaker is labeled 40% salt solution and the solution inside the cell is labeled 20% salt solution.only water is allowed to move in and out of the cell. what will most likely happen to the cell? it will expand as water moves out of it. it will shrink as water moves out of it.it will expand as water moves into it. it will shrink as water moves into it.
Answers: 2

Chemistry, 22.06.2019 12:30
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

You know the right answer?
Calculate the pH of a 0.0255 M solution of ammonium bromide (NH4Br). The Kb of ammonia is 1.76 x 10-...
Questions

Mathematics, 04.06.2020 21:01





Mathematics, 04.06.2020 21:01




Chemistry, 04.06.2020 21:02

Mathematics, 04.06.2020 21:02

Chemistry, 04.06.2020 21:02

Mathematics, 04.06.2020 21:02

Mathematics, 04.06.2020 21:02

Chemistry, 04.06.2020 21:02

Mathematics, 04.06.2020 21:02

English, 04.06.2020 21:02



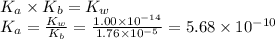
![[H_3O^{+} ]= \sqrt{K_a \times C_a } = \sqrt{5.68 \times 10^{-10} \times 0.0255 } = 3.81 \times 10^{-6}M](/tpl/images/0705/0989/d386c.png)
![pH = -log [H_3O^{+} ] = -log (3.81 \times 10^{-6}) = 5.42](/tpl/images/0705/0989/96bbf.png)


