

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:40
It is important to wear proper protective equipment in lab even when not actively performing experiments because accidents can affect any researcher, even one not working on an experiment. select the best answer from the choices provided
Answers: 3

Chemistry, 21.06.2019 23:30
Calculate the expected ph values of the buffer systems from the experiments (a,b,c,d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2

Chemistry, 22.06.2019 07:00
Which set of characteristics best describes igneous rock? a) largest type of rock, made of organic matter, hardest type of rock b) least abundant type of rock, made of other rocks, made mostly of minerals c) found on all continents, contains wavy bands of stripes, contains fossils d) most abundant type in earth's crust, made of magma/lava, contains no fossils
Answers: 1

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1
You know the right answer?
What is the equilibrium concentration of ICl if 0.45 mol of I2 and 0.45 mol of Cl2 are initially mix...
Questions



History, 04.09.2020 20:01


Mathematics, 04.09.2020 20:01

Mathematics, 04.09.2020 20:01

Health, 04.09.2020 20:01



Mathematics, 04.09.2020 20:01


Mathematics, 04.09.2020 20:01



Mathematics, 04.09.2020 20:01


English, 04.09.2020 20:01

Mathematics, 04.09.2020 20:01


Physics, 04.09.2020 20:01

![[ICI]_{eq}=0.271M](/tpl/images/0705/0745/27a40.png)
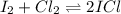
![[I_2]_0=[Cl_2]_0=\frac{0.45mol}{2.0L}= 0.225M](/tpl/images/0705/0745/f16cc.png)
 due to the reaction extent:
due to the reaction extent:![Kc=\frac{[ICI]^2}{([I_2])([CI_2])} \\\\Kc=\frac{(2x)^2}{([I_2]_0-x)([CI_2]_0-x)} \\\\9.09=\frac{(2x)^2}{(0.225-x)(0.225-x)}](/tpl/images/0705/0745/ac0ef.png)
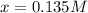
![[ICI]_{eq}=2*0.135M](/tpl/images/0705/0745/888be.png)


