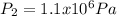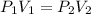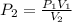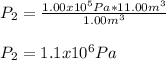Chemistry, 15.07.2020 03:01 EbonyMontricee
an ideal gas is at a pressure 1.00 x 10^5 N/m^2 and occupies a volume 11.00 m^3. If the gass is compressed to a volume of 1.00 m^3 while the temperature remains constant, what will be the new pressure in the gas.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 20:30
Citric acid has a ph between 1 and 3. it is considered to be aa. weak acidb. weak basec. strong based. strong acid
Answers: 2

Chemistry, 23.06.2019 00:30
If there are 3.5 moles of koh, how many moles of naoh can be produced? question 1 options: a)3.0 moles naoh b)3.5 moles naoh c)1 moles naoh d)9 moles naoh
Answers: 1

Chemistry, 23.06.2019 04:20
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1

Chemistry, 23.06.2019 19:00
X-rays are used instead of visible light because x-rays have shorter than visible light, allowing them to produce images of much greater detail. a) energies b) frequencies c) speeds d) wavelengths
Answers: 3
You know the right answer?
an ideal gas is at a pressure 1.00 x 10^5 N/m^2 and occupies a volume 11.00 m^3. If the gass is comp...
Questions

Mathematics, 27.06.2019 15:00

Mathematics, 27.06.2019 15:00


Mathematics, 27.06.2019 15:00






History, 27.06.2019 15:00






Mathematics, 27.06.2019 15:00


History, 27.06.2019 15:00

Mathematics, 27.06.2019 15:00