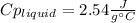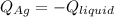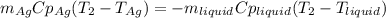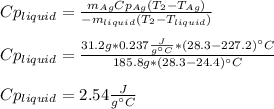Chemistry, 16.07.2020 01:01 makaylaf9479
3. A 31.2-g piece of silver (s = 0.237 J/(g · °C)), initially at 277.2°C, is added to 185.8 g of a liquid, initially at 24.4°C, in an insulated container. The final temperature of the metal–liquid mixture at equilibrium is 28.3°C. What is the specific heat of the liquid? Neglect the heat capacity of the container.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
14. complete and balance the equations for the single displacement reactions. a. zn + pb(no3)2 -> b. al + niso4 -> 15. complete and balance the equations for the double displacement reactions. a. agno3(aq) + nacl(aq) -> b. mg(no3)2(aq) + koh(aq) -> 16. complete and balance the equations for the combustion reactions. a. __ ch4 + o2 -> b. __ c3h6 + o2 -> c. + o2 ->
Answers: 2

Chemistry, 22.06.2019 15:20
An alloy contains 66 g of pure zinc. what is the percentage of zinc in the alloy? express your answer to two significant figures and include the appropriate units.
Answers: 1

Chemistry, 23.06.2019 03:00
Give a real-world example of an energy transformation that uses two of the following forms of energy: chemical, mechanical, nuclear, gravitational, radiant, electrical, thermal (heat), and/or sound.
Answers: 3

Chemistry, 23.06.2019 03:00
Which of the following is a chemical property of water at 4 c
Answers: 2
You know the right answer?
3. A 31.2-g piece of silver (s = 0.237 J/(g · °C)), initially at 277.2°C, is added to 185.8 g of a l...
Questions

Mathematics, 10.09.2021 01:00


Biology, 10.09.2021 01:00

Mathematics, 10.09.2021 01:00

Chemistry, 10.09.2021 01:00





Mathematics, 10.09.2021 01:00

Biology, 10.09.2021 01:00

Mathematics, 10.09.2021 01:00


Mathematics, 10.09.2021 01:00

Mathematics, 10.09.2021 01:00


Mathematics, 10.09.2021 01:00

Mathematics, 10.09.2021 01:00


Engineering, 10.09.2021 01:00