
Chemistry, 16.07.2020 02:01 marykm03p3sd80
3. A student carries out the clay-catalyzed dehydration of cyclohexanol starting with 10 moles of cyclohexanol and obtains 500 mL of pure cyclohexene product. Calculate the student’s percent yield for this reaction. Show your work.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Will mark brainliest26. which of these statements are true? (3 points)a. gases are compressibleb. gases fill their containers completelyc. the pressure of a gas is independent of the temperatured. gases have masse. gases exert pressuref. the pressure of a gas is dependent on the volumeg. gas pressure results from the collisions between gas particlesh. gases have a definite volume and shape
Answers: 1

Chemistry, 22.06.2019 00:20
What are the spectator ions in 2h+ + so42- + ca2+ + 2r → caso4 + 2h+ + 21?
Answers: 1

Chemistry, 22.06.2019 13:30
What does the xylem do? stores the glucose captures the sunlight absorbs oxygen into the leaf carries water from the roots to the leaves
Answers: 1

Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1
You know the right answer?
3. A student carries out the clay-catalyzed dehydration of cyclohexanol starting with 10 moles of cy...
Questions

Mathematics, 25.09.2019 11:50






English, 25.09.2019 11:50

English, 25.09.2019 11:50



History, 25.09.2019 11:50

Computers and Technology, 25.09.2019 11:50


Spanish, 25.09.2019 11:50

Mathematics, 25.09.2019 11:50

Chemistry, 25.09.2019 11:50

History, 25.09.2019 11:50

History, 25.09.2019 11:50

Geography, 25.09.2019 11:50

History, 25.09.2019 11:50

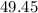
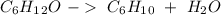
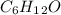 ) we will obtain 10 mol of cyclohexanol (
) we will obtain 10 mol of cyclohexanol (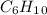 ). So, we can calculate the grams of cyclohexanol if we calculate the molar mass:
). So, we can calculate the grams of cyclohexanol if we calculate the molar mass:

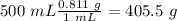
 %= (405.5/820)*100 = 49.45 %
%= (405.5/820)*100 = 49.45 %

