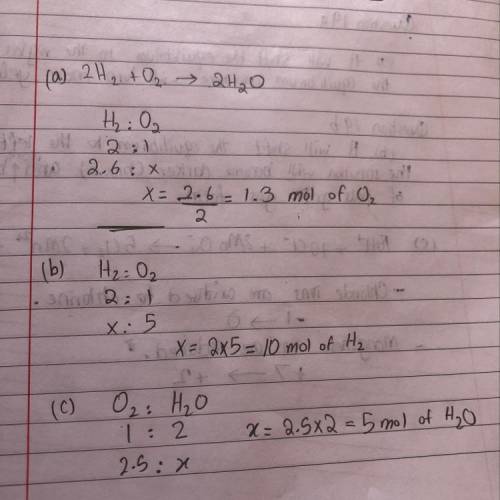The reaction of hydrogen with oxygen produces water.
2H2(g) + O2(8) - 2H2O(g)
a. How many mol...

The reaction of hydrogen with oxygen produces water.
2H2(g) + O2(8) - 2H2O(g)
a. How many moles of O2 are required to react with 2.6 mol
of H2?
b. How many moles of H, are needed to react with 5.0 mol
of O2?
c. How many moles of H2O form when 2.5 mol of O2
reacts?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Write the empirical chemical formula of calcium with a mass percent of 38.8, phosphorus with a mass percent of 20.0, and oxygen with a mass percent of 41.3.
Answers: 1

Chemistry, 22.06.2019 04:00
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3


You know the right answer?
Questions

Computers and Technology, 23.07.2019 22:00



History, 23.07.2019 22:00

Mathematics, 23.07.2019 22:00

Mathematics, 23.07.2019 22:00

Mathematics, 23.07.2019 22:00



Biology, 23.07.2019 22:00


Biology, 23.07.2019 22:00


History, 23.07.2019 22:00




Mathematics, 23.07.2019 22:00

Mathematics, 23.07.2019 22:00