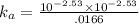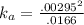Chemistry, 23.07.2020 20:01 brendancrow5927
Enough of a monoprotic weak acid is dissolved in water to produce a 0.01660.0166 M solution. The pH of the resulting solution is 2.532.53 . Calculate the Ka for the acid.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Which of these is not an example of chemical weathering? a. iron-rich mineral rusting b. feldspar turning into clay c. limestone reacting with acid d. granite breaking up into sand
Answers: 1

Chemistry, 22.06.2019 10:30
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1


Chemistry, 23.06.2019 01:00
Chromium(iii) sulfate is a transition metal compound containing the metal chromium and the polyatomic ion sulfate. the oxidation state of chromium in this compound is , and the chemical formula of the compound is ( ) . reset next
Answers: 3
You know the right answer?
Enough of a monoprotic weak acid is dissolved in water to produce a 0.01660.0166 M solution. The pH...
Questions







English, 25.09.2019 06:00

Physics, 25.09.2019 06:00


English, 25.09.2019 06:00



History, 25.09.2019 06:00


Mathematics, 25.09.2019 06:00


Physics, 25.09.2019 06:00



Mathematics, 25.09.2019 06:00

![[ H^+]=10^{-2.53}](/tpl/images/0711/9511/3a08a.png)
![[ X^-]=10^{-2.53}](/tpl/images/0711/9511/d1b85.png)