Approximately 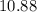 .
.
Explanation:
Equilibrium constant
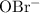 can act as a weak Bronsted-Lowry base:
can act as a weak Bronsted-Lowry base:
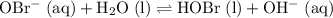 .
.
(Side note: the state symbol of 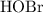 in this equation is
in this equation is 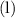 (meaning liquid) because
(meaning liquid) because 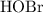 is a weak acid.)
is a weak acid.)
However, the equilibrium constant of this reaction, 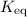 , isn't directly given. The idea is to find
, isn't directly given. The idea is to find 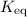 using the
using the 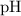 value at the half-equivalence point. Keep in mind that this system is at equilibrium all the time during the titration. If temperature stays the same, then the same
value at the half-equivalence point. Keep in mind that this system is at equilibrium all the time during the titration. If temperature stays the same, then the same 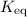 value could also be used to find the
value could also be used to find the 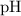 of the solution before the acid was added.
of the solution before the acid was added.
At equilibrium:
![\displaystyle K_\text{eq} = \frac{[\rm HOBr\; (l)]\cdot [\rm OH^{-}\; (aq)]}{[\rm OBr^{-}\; (aq)]}](/tpl/images/0711/7921/8c12d.png) .
.
At the half-equivalence point of this titration, exactly half of the base, 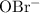 , has been converted to its conjugate acid,
, has been converted to its conjugate acid, 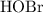 . Therefore, the half-equivalence concentration of
. Therefore, the half-equivalence concentration of 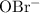 and
and 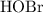 should both be equal to one-half the initial concentration of
should both be equal to one-half the initial concentration of 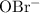 .
.
As a result, the half-equivalence concentration of 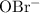 and
and 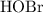 should be the same. The expression for
should be the same. The expression for 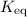 can thus be simplified:
can thus be simplified:
![\begin{aligned}& K_\text{eq} \\&= \frac{\left(\text{half-equivalence $[\rm HOBr\; (l)]$}\right)\cdot \left(\text{half-equivalence $[\rm OH^{-}\; (aq)]$}\right)}{\text{half-equivalence $[\rm OBr^{-}\; (l)]$}}\\ &=\text{half-equivalence $[\rm OH^{-}\; (aq)]$}\end{aligned}](/tpl/images/0711/7921/4e833.png) .
.
In other words, the 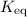 of this system is equal to the
of this system is equal to the 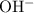 concentration at the half-equivalence point. Assume that
concentration at the half-equivalence point. Assume that 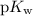 the self-ionization constant of water, is
the self-ionization constant of water, is 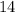 . The concentration of
. The concentration of 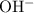 can be found from the
can be found from the 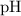 value:
value:
![\begin{aligned}& \text{half-equivalence $[\rm OH^{-}\; (aq)]$} \\ &= 10^{\rm pH - p\mathnormal{K}_\text{w}}\;\rm mol \cdot L^{-1} \\ &= 10^{7.75 - 14}\; \rm mol \cdot L^{-1}\\ &= 10^{-6.25}\; \rm mol \cdot L^{-1}\end{aligned}](/tpl/images/0711/7921/38e0b.png) .
.
Therefore, 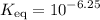 .
.
Initial pH of the solution
Again, since 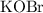 is a soluble salt, all that
is a soluble salt, all that 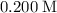 of
of 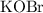 in this solution will be in the form of
in this solution will be in the form of 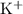 and
and 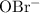 ions. Before any hydrolysis takes place, the concentration of
ions. Before any hydrolysis takes place, the concentration of 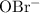 should be equal to that of
should be equal to that of 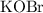 . Therefore:
. Therefore:
![\text{$[\rm OBr^{-}\; (aq)]$ before hydrolysis} = 0.200\; \rm M](/tpl/images/0711/7921/ea399.png) .
.
Let the equilibrium concentration of ![[\rm OH^{-}\; (aq)]](/tpl/images/0711/7921/bc9d8.png) be
be 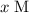 . Create a RICE table for this reversible reaction:
. Create a RICE table for this reversible reaction:
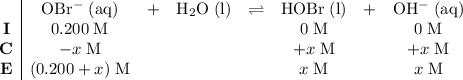 .
.
Assume that external factors (such as temperature) stays the same. The 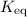 found at the half-equivalence point should apply here, as well.
found at the half-equivalence point should apply here, as well.
![\displaystyle K_\text{eq} = \frac{[\rm HOBr\; (l)]\cdot [\rm OH^{-}\; (aq)]}{[\rm OBr^{-}\; (aq)]}](/tpl/images/0711/7921/8c12d.png) .
.
At equilibrium:
![\displaystyle \frac{[\rm HOBr\; (l)]\cdot [\rm OH^{-}\; (aq)]}{[\rm OBr^{-}\; (aq)]} = \frac{x^2}{0.200 + x}](/tpl/images/0711/7921/e2d3c.png) .
.
Assume that  is much smaller than
is much smaller than 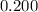 , such that the denominator is approximately the same as
, such that the denominator is approximately the same as 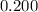 :
:
![\displaystyle \frac{[\rm HOBr\; (l)]\cdot [\rm OH^{-}\; (aq)]}{[\rm OBr^{-}\; (aq)]} = \frac{x^2}{0.200 + x} \approx \frac{x^2}{0.200}](/tpl/images/0711/7921/f87a1.png) .
.
That should be equal to the equilibrium constant, 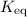 . In other words:
. In other words:
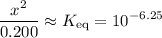 .
.
Solve for  :
:
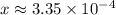 .
.
In other words, the 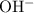 before acid was added was approximately
before acid was added was approximately 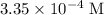 , which is the same as
, which is the same as 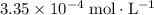 . Again, assume that
. Again, assume that 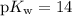 . Calculate the
. Calculate the 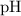 of that solution:
of that solution:
![\begin{aligned}\rm pH &= \rm p\mathnormal{K}_\text{w} + \log [\mathrm{OH^{-}}] \approx 10.88\end{aligned}](/tpl/images/0711/7921/8add0.png) .
.
(Rounded to two decimal places.)


























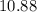 .
.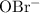 can act as a weak Bronsted-Lowry base:
can act as a weak Bronsted-Lowry base: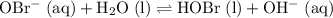 .
.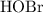 in this equation is
in this equation is 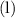 (meaning liquid) because
(meaning liquid) because 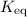 , isn't directly given. The idea is to find
, isn't directly given. The idea is to find 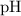 value at the half-equivalence point. Keep in mind that this system is at equilibrium all the time during the titration. If temperature stays the same, then the same
value at the half-equivalence point. Keep in mind that this system is at equilibrium all the time during the titration. If temperature stays the same, then the same ![\displaystyle K_\text{eq} = \frac{[\rm HOBr\; (l)]\cdot [\rm OH^{-}\; (aq)]}{[\rm OBr^{-}\; (aq)]}](/tpl/images/0711/7921/8c12d.png) .
.![\begin{aligned}& K_\text{eq} \\&= \frac{\left(\text{half-equivalence $[\rm HOBr\; (l)]$}\right)\cdot \left(\text{half-equivalence $[\rm OH^{-}\; (aq)]$}\right)}{\text{half-equivalence $[\rm OBr^{-}\; (l)]$}}\\ &=\text{half-equivalence $[\rm OH^{-}\; (aq)]$}\end{aligned}](/tpl/images/0711/7921/4e833.png) .
. 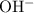 concentration at the half-equivalence point. Assume that
concentration at the half-equivalence point. Assume that 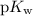 the self-ionization constant of water, is
the self-ionization constant of water, is 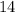 . The concentration of
. The concentration of ![\begin{aligned}& \text{half-equivalence $[\rm OH^{-}\; (aq)]$} \\ &= 10^{\rm pH - p\mathnormal{K}_\text{w}}\;\rm mol \cdot L^{-1} \\ &= 10^{7.75 - 14}\; \rm mol \cdot L^{-1}\\ &= 10^{-6.25}\; \rm mol \cdot L^{-1}\end{aligned}](/tpl/images/0711/7921/38e0b.png) .
.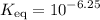 .
. is a soluble salt, all that
is a soluble salt, all that 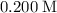 of
of  and
and ![\text{$[\rm OBr^{-}\; (aq)]$ before hydrolysis} = 0.200\; \rm M](/tpl/images/0711/7921/ea399.png) .
.![[\rm OH^{-}\; (aq)]](/tpl/images/0711/7921/bc9d8.png) be
be 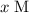 . Create a RICE table for this reversible reaction:
. Create a RICE table for this reversible reaction: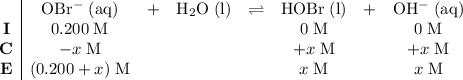 .
.![\displaystyle \frac{[\rm HOBr\; (l)]\cdot [\rm OH^{-}\; (aq)]}{[\rm OBr^{-}\; (aq)]} = \frac{x^2}{0.200 + x}](/tpl/images/0711/7921/e2d3c.png) .
. is much smaller than
is much smaller than 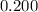 , such that the denominator is approximately the same as
, such that the denominator is approximately the same as ![\displaystyle \frac{[\rm HOBr\; (l)]\cdot [\rm OH^{-}\; (aq)]}{[\rm OBr^{-}\; (aq)]} = \frac{x^2}{0.200 + x} \approx \frac{x^2}{0.200}](/tpl/images/0711/7921/f87a1.png) .
.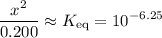 .
.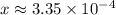 .
.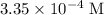 , which is the same as
, which is the same as 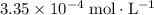 . Again, assume that
. Again, assume that 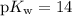 . Calculate the
. Calculate the ![\begin{aligned}\rm pH &= \rm p\mathnormal{K}_\text{w} + \log [\mathrm{OH^{-}}] \approx 10.88\end{aligned}](/tpl/images/0711/7921/8add0.png) .
. 

