Chemistry, 25.07.2020 06:01 NikolaiSolokov
You are given 10.00 mL of a solution of an unknown acid. The pH of this solution is exactly 2.18. You determine that the concentration of the unknown acid was 0.2230 M. You also determined that the acid was monoprotic (HA). What is the pKa of your unknown acid

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:00
Select all that apply. a beta particle: is electromagnetic energy is an electron has zero charge is emitted from the nucleus has a +2 charge has a -1 charge
Answers: 1

Chemistry, 22.06.2019 03:50
Express the following number in scientific notation. 0.026890 =
Answers: 1


Chemistry, 22.06.2019 19:00
Convert the temperature of dry ice, –77 ∞c, into degrees fahrenheit and kelvin.
Answers: 2
You know the right answer?
You are given 10.00 mL of a solution of an unknown acid. The pH of this solution is exactly 2.18. Yo...
Questions

Advanced Placement (AP), 28.01.2021 03:30

Biology, 28.01.2021 03:30

Mathematics, 28.01.2021 03:30


English, 28.01.2021 03:30

Mathematics, 28.01.2021 03:30

Mathematics, 28.01.2021 03:30

Health, 28.01.2021 03:30


Mathematics, 28.01.2021 03:30






Mathematics, 28.01.2021 03:30



Mathematics, 28.01.2021 03:30

History, 28.01.2021 03:30

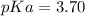
![pH=-log([H^+])\\](/tpl/images/0712/8744/a9ef9.png)
![[H^+]=10^{-pH}=10^{-2.18}=6.61x10^{-3}M](/tpl/images/0712/8744/456e0.png)
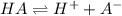
![Ka=\frac{[H^+][A^-]}{[HA]}](/tpl/images/0712/8744/39962.png)
![[HA]=0.2230M-6.61x10^{-3}M=0.2164M](/tpl/images/0712/8744/1a456.png)
 ). Thereby, the acid dissociation constant turns out:
). Thereby, the acid dissociation constant turns out: