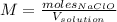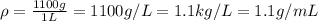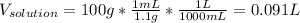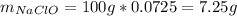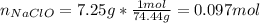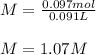Chemistry, 27.07.2020 01:01 santana647
1L of bleach has a mass of 1,100 grams, 7.25% of the mass of bleach is NaClO, 1 mol of NaClO has a mass of 74.44 grams. What is the molarity (mol/L) of NaClO in the bleach? A.0.097 B.0.93 C. 1.07 D.79.75

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 14:30
An occluded front moves over the farmland that has been experiencing drought conditions. what change in weather will this front likely bring? a. gray skies, but no rain b. an extended period of rain c. more dry air and sunny skies d. violent, short-lived thunderstorms
Answers: 3

Chemistry, 21.06.2019 14:30
As a part of an experiment a student burns propane to produce carbon dioxide and water she remembers that she must follow the law conservation of matter when writing a balanced chemical equation which of these equation adheres to the law of conservation of matter
Answers: 1


Chemistry, 22.06.2019 04:00
The continuous release of nuclear energy caused when one fission reaction triggered more nuclear reactions is a
Answers: 3
You know the right answer?
1L of bleach has a mass of 1,100 grams, 7.25% of the mass of bleach is NaClO, 1 mol of NaClO has a m...
Questions



Mathematics, 10.12.2020 06:20




Mathematics, 10.12.2020 06:20

Mathematics, 10.12.2020 06:20

Spanish, 10.12.2020 06:20



Mathematics, 10.12.2020 06:20

Mathematics, 10.12.2020 06:20




Mathematics, 10.12.2020 06:20