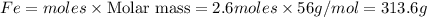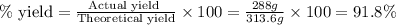Chemistry, 27.07.2020 02:01 stefkellum58
Fe2O3(s) + 3CO(g) ---> 2Fe(l) + 3CO2(g) Steve inserts 450. g of iron(III) oxide and 260. g of carbon monoxide into the blast furnace. After cooling the pure liquid iron, Steve determines that he has produced 288g of iron ingots. Use the theoretical yield of liquid iron and the mass or iron ingots to calculate the percent yield of the reaction.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Using the periodic table, complete the table to describe each atom. type in your answers
Answers: 3

Chemistry, 22.06.2019 15:20
An alloy contains 66 g of pure zinc. what is the percentage of zinc in the alloy? express your answer to two significant figures and include the appropriate units.
Answers: 1

Chemistry, 22.06.2019 20:00
Glucose (c6h12o6) is an important biological molecule. (round the answer to nearest hundredth.) what is the percent by mass of carbon in glucose?
Answers: 2

Chemistry, 23.06.2019 05:00
He nucleus contains the cells genetic material in the form of dna. dna is organized into our chromosomes, which are made up of thousands of that determine our traits.
Answers: 1
You know the right answer?
Fe2O3(s) + 3CO(g) ---> 2Fe(l) + 3CO2(g) Steve inserts 450. g of iron(III) oxide and 260. g of car...
Questions


Mathematics, 03.11.2019 07:31


Mathematics, 03.11.2019 07:31



Mathematics, 03.11.2019 07:31

Spanish, 03.11.2019 07:31

Mathematics, 03.11.2019 07:31


History, 03.11.2019 07:31

Biology, 03.11.2019 07:31

Mathematics, 03.11.2019 07:31




Chemistry, 03.11.2019 07:31


Mathematics, 03.11.2019 07:31

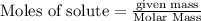
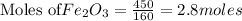
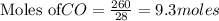
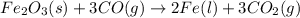
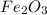 require 3 moles of
require 3 moles of 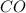
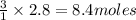 of
of 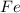
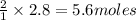 of
of