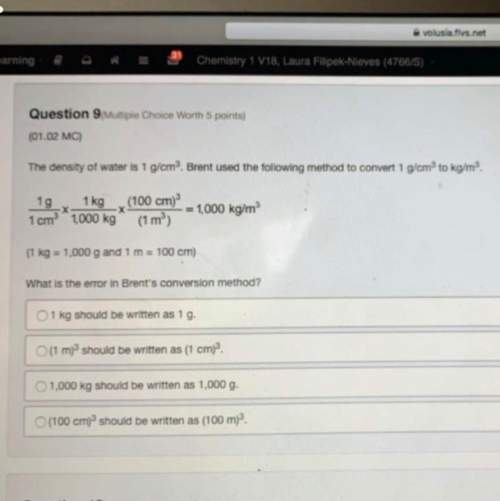
Mass of flask acid= 98.788
Mass of flask = 98.318
Mass of weak acid???
What is th...

Chemistry, 27.07.2020 23:01 rgilliam3002
Mass of flask acid= 98.788
Mass of flask = 98.318
Mass of weak acid???
What is the mass of weak acid?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:00
Select all that apply. a beta particle: is electromagnetic energy is an electron has zero charge is emitted from the nucleus has a +2 charge has a -1 charge
Answers: 1

Chemistry, 22.06.2019 04:30
Which statement best describes the relationship between period and frequency of light waves? a) in wave b the period increases and the frequency decreases from wave a. b) in wave a the period increases and the frequency decreases from wave b. c) in wave b the period is shorter and the frequency is greater than in wave a. d) in wave a the period is shorter and the frequency is greater than in wave b.
Answers: 1

Chemistry, 22.06.2019 08:30
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3

Chemistry, 22.06.2019 12:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al oxidizing agent = reducing agent =
Answers: 1
You know the right answer?
Questions



Mathematics, 01.11.2020 17:50




English, 01.11.2020 17:50


Business, 01.11.2020 17:50

Mathematics, 01.11.2020 17:50



Social Studies, 01.11.2020 17:50

Mathematics, 01.11.2020 17:50

World Languages, 01.11.2020 17:50

Spanish, 01.11.2020 17:50


English, 01.11.2020 17:50

Mathematics, 01.11.2020 17:50

English, 01.11.2020 17:50