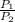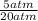Chemistry, 29.07.2020 05:01 miannasanderso8146
2) 2.5 mol of an ideal gas at 20 oC under 20 atm pressure, was expanded up to 5 atm pressure via; (a) adiabatic reversible and (b) adiabatic irreversible process. Calculate the values of w, q, ΔU, ΔH for each process. (Cv = 5 cal / mol. K ≈ 5/2 R; R ≈ 2 cal / mol. K) (Please find the desired values by making the corresponding derivations

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which statement correctly describes potassium iodide, ki? there is a one-to-one ratio of potassium ions to iodide ions. potassium gains electrons and iodine loses electrons during the reaction. the lattice is held together by potassium anions and iodide cations.
Answers: 1

Chemistry, 22.06.2019 02:10
What approach is required to balance the objectives of sustainable development? balancing the objectives of sustainable development requires a(n) .
Answers: 3

Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be changed varied experimented controlled
Answers: 1
You know the right answer?
2) 2.5 mol of an ideal gas at 20 oC under 20 atm pressure, was expanded up to 5 atm pressure via; (a...
Questions






English, 26.09.2019 04:10

English, 26.09.2019 04:10






Mathematics, 26.09.2019 04:10


Mathematics, 26.09.2019 04:10

History, 26.09.2019 04:10

Arts, 26.09.2019 04:10

Mathematics, 26.09.2019 04:10


Mathematics, 26.09.2019 04:10