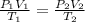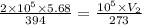Chemistry, 30.07.2020 05:01 andresduenas72
A pressure cooker contains 5.68 liters of air at a temperature of 394 K. If the absolute pressure of the air in the pressure cooker is 205 pascals, how many moles of air are in the cooker? The cooker contains moles of air. 1 SEE ANSWER

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 12:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1

Chemistry, 22.06.2019 19:00
How does kepler second law of planetary motion overthrow one of the basic beliefs of classical astronomy
Answers: 1

Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1
You know the right answer?
A pressure cooker contains 5.68 liters of air at a temperature of 394 K. If the absolute pressure of...
Questions



English, 08.03.2021 07:30

Biology, 08.03.2021 07:30

Mathematics, 08.03.2021 07:30






Mathematics, 08.03.2021 07:30


Mathematics, 08.03.2021 07:30

Mathematics, 08.03.2021 07:30



Mathematics, 08.03.2021 07:30


Mathematics, 08.03.2021 07:30

Mathematics, 08.03.2021 07:30