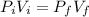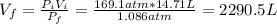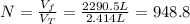A cylinder containing 14.71 L of helium gas at a pressure of 169.1 atm is to be used to fill toy balloons to a pressure of 1.086 atm. Each inflated balloon has a volume of 2.414 L. What is the maximum number of balloons that can be inflated? Report your answer to 1 decimal place. (Remember that 14.71 L of helium at 1.086 atm will remain in the exhausted (empty) cylinder)

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 17:50
Cryolite, na3alf6(s), an ore used in the production of aluminum, can be synthesized using aluminum oxide. start this question by first balance the chemical equation.1.) balance the equation: - alo3(s)+naoh(l)+hf(> na3alf6+h2o(g). 2.) if 17.5 kilograms of al2o3(s), 51.4 kilograms of naoh(l), and 51.4 kilograms of hf(g) react completely, how many kilograms of cryolite will be produced? 3.)which reactants will be in excess, (al2o3, naoh, or hf) 4.)what is the total mass of the excess reactants left over after the reaction is complete in kg?
Answers: 2

Chemistry, 22.06.2019 18:20
Categorize them by metal, nonmetal, in periodic tableductilenon-ductilemalleableoften gain electrons easilygood conductorpoor conductorcan be liquidselements
Answers: 2

Chemistry, 22.06.2019 21:00
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3
You know the right answer?
A cylinder containing 14.71 L of helium gas at a pressure of 169.1 atm is to be used to fill toy bal...
Questions

Mathematics, 29.01.2020 11:49

English, 29.01.2020 11:49

Mathematics, 29.01.2020 11:49

Chemistry, 29.01.2020 11:49

History, 29.01.2020 11:49

English, 29.01.2020 11:49


Mathematics, 29.01.2020 11:49

Computers and Technology, 29.01.2020 11:49



Mathematics, 29.01.2020 11:49

History, 29.01.2020 11:49



Mathematics, 29.01.2020 11:49

Mathematics, 29.01.2020 11:49



Social Studies, 29.01.2020 11:49

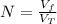
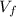 : is the volume at 1.086 atm
: is the volume at 1.086 atm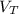 : is the balloon volume = 2.414 L
: is the balloon volume = 2.414 L