
Chemistry, 04.08.2020 23:01 elijahjacksonrp6z2o7
Part 1. Determine the molar mass of a 0.622-gram sample of gas having a volume of 2.4 L at 287 K and 0.850 atm. Show your work. Part 2. If this sample was placed under extremely low temperature, describe how the actual volume would compare to the predicted volume. Explain your answer.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 16:50
Ajet plane is speeding down the runway during takeoff. air resistance is not negligible. identify the forces on the jet.
Answers: 3

Chemistry, 22.06.2019 21:30
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1

Chemistry, 23.06.2019 00:10
Covalent compounds: mastery test select the correct answer what is formed when atoms join together with a covalent bond? a. an ion b. a molecule c. a neutral atom d. a noble gas
Answers: 3
You know the right answer?
Part 1. Determine the molar mass of a 0.622-gram sample of gas having a volume of 2.4 L at 287 K and...
Questions


Mathematics, 10.11.2019 13:31




English, 10.11.2019 13:31

Biology, 10.11.2019 13:31


Biology, 10.11.2019 13:31

Mathematics, 10.11.2019 13:31


History, 10.11.2019 13:31

Geography, 10.11.2019 13:31

Mathematics, 10.11.2019 13:31

History, 10.11.2019 13:31



Mathematics, 10.11.2019 13:31


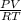
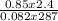
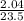 = 0.087mole
= 0.087mole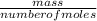
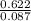 =7.15gmol⁻¹
=7.15gmol⁻¹

